LIPS (Ligand Induced Phase Shift) is an innovative, label-free method developed to evaluate functional target engagement within disease models. By leveraging the intrinsic changes in a protein’s physicochemical properties upon drug binding, LIPS offers a sensitive and quantitative way to monitor how drugs interact with their targets in both cultured cell lines and diseased human tissues.
LIPS rationale:

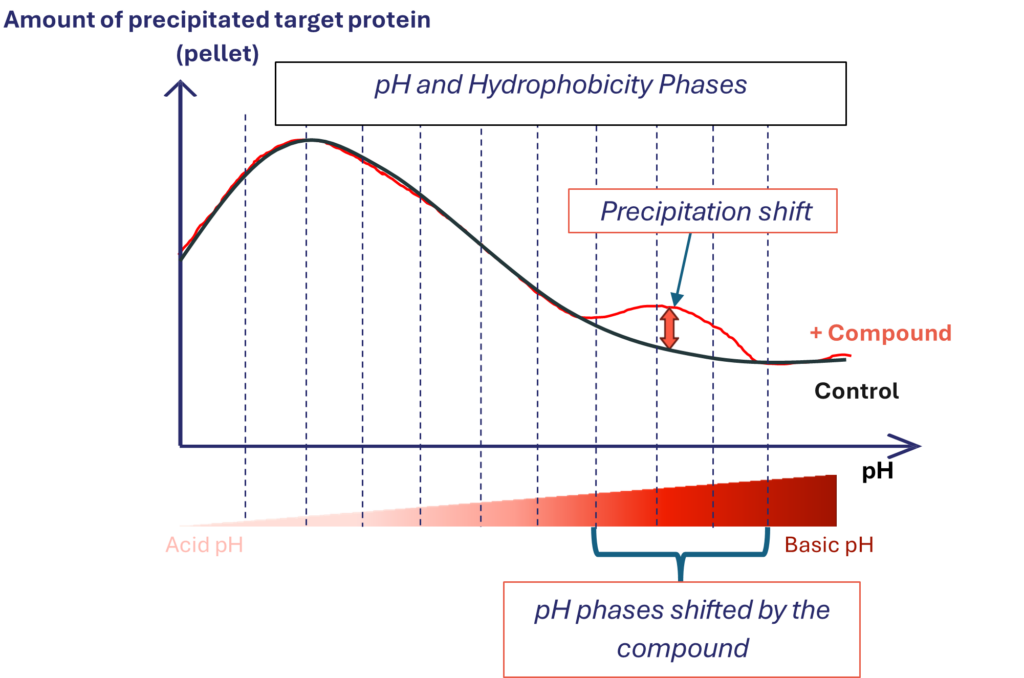
LIPS is based on the shift in charge distribution of target upon drug-target engagement along microscopic pH and hydrophobicity gradients. It is used to show dose-dependent drug-target engagement in complex tissue or cell line both in vitro and ex vivo
How it works
Drug treatment and sample preparation
- Dose- and time-dependent exposure: Cells or patient-derived tissues are treated with increasing concentrations of a drug over various time points. This approach not only reveals dose-dependent effects but also captures the kinetics of target engagement, providing insights into how quickly and robustly the target responds to the drug.
- Cell lysis: After treatment, the cells are lysed to release their protein contents.
- Gradient plating: The lysate is then distributed across plates featuring finely tuned pH and hydrophobicity gradients. This setup is crucial because drug binding alters the target protein’s charge distribution and isoelectric point (pI).
Phase shift mechanism
- Physicochemical alterations: When a drug engages its target, it induces a shift in the protein’s charge distribution. This change affects how the protein behaves under varying pH and hydrophobic conditions—specifically, its tendency to precipitate.
- Phase Separation: As a result, the target protein partitions differently between precipitated (pellet) and soluble (supernatant) fractions. An increase in drug concentration or prolonged exposure typically leads to a higher proportion of the target protein precipitating, reflecting enhanced and time-dependent drug-target engagement.
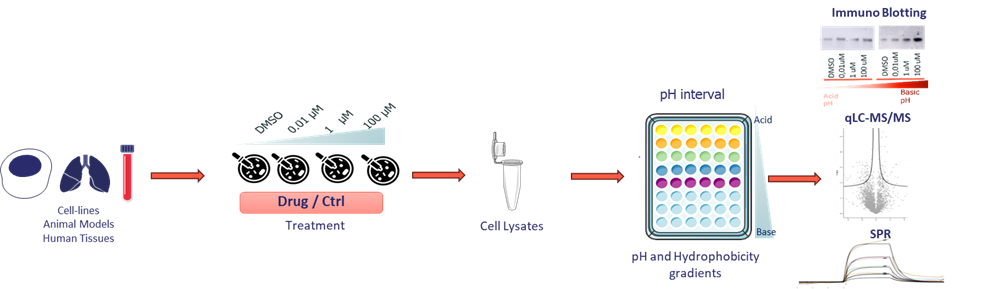
Multi-layered readouts
To comprehensively assess these changes, LIPS integrates three advanced analytical techniques:
Immunoblotting (WB):
- Isoelectric point shift detection: WB monitors changes in the target’s immunoreactivity, particularly shifts in its isoelectric point. By comparing the intensity of the protein bands in WB across different fractions and treatment conditions, we can infer that the protein’s physicochemical properties have been altered. If higher drug concentrations (or longer treatment times) result in more protein in fractions corresponding to a particular pH, it suggests a drug-induced shift in the protein’s pI.
- Quantitative comparison: At higher drug concentrations or longer treatment times, an increased target protein presence in the pellet (and a corresponding decrease in the supernatant) confirms dose- and time-dependent engagement.
Label-Free Quantitative Proteomics (LC-MS/MS):
- Protein enrichment profiling: This approach quantitatively analyses proteins in both pellet and supernatant fractions.
- Network analysis: It not only identifies the target protein but also captures changes in its interacting partners, shedding light on broader protein network modulations induced by the drug.
Surface Plasmon Resonance (SPR):
- Kinetic and specificity insights: SPR provides real-time measurements of the direct interactions between the drug and its target, delivering essential data on binding kinetics (e.g., dissociation constant, Kd).
- Validation of engagement: It also assesses the specificity, sensitivity, and selectivity of the drug, thereby validating the functional relevance of the observed target engagement.
Applications and advantages
Versatility
Applicable in both in vitro (cell lines) and ex vivo (patient tissue) settings, making it a powerful tool for translational research.
Label-free
By eliminating the need for fluorescent or radioactive labels, LIPS preserves the native state of the proteins, ensuring more physiologically relevant data.
Comprehensive analysis
The integration of WB, quantitative proteomics, and SPR provides a multi-dimensional view of drug-target interactions—from molecular alterations and dose-dependent responses to kinetic properties across different time points.
In summary, LIPS offers a robust, quantitative, and label-free method for assessing dose- and time-dependent drug-target engagement. This approach enhances our understanding of the underlying molecular mechanisms in disease models and supports the development and validation of therapeutic compounds with high specificity and efficacy.