Physiological Intermolecular Modulation Spectroscopy (PIMS®) is a patented, label-free technology that uses near-infrared (NIR) spectroscopy to probe the interactions between therapeutic agents (e.g., drugs, peptides, biologics) and their molecular targets (e.g., proteins) in a patient’s biological sample. Unlike traditional assays that often require fluorescent or radioactive labeling, PIMS® exploits water molecule resonance as a direct indicator of molecular changes, preserving the sample’s native state.
Parallels with MRI
A useful analogy is Magnetic Resonance Imaging (MRI), a technology that also relies on water as a proxy for structural and functional changes in tissues. However, the physical principles differ:
MRI measures the relaxation times (T1, T2) of hydrogen nuclei in water under a strong magnetic field and radiofrequency pulses. This provides anatomical and functional images of tissue on a macroscopic scale.
PIMS®, in contrast, operates in the near-infrared (NIR) range (approximately 970–1940 nm) to detect changes in water absorption at a molecular scale. Instead of creating an anatomical image, PIMS® delivers a spectral fingerprint that reflects how a specific compound (drug, antibody, peptide) shifts the water network in its immediate vicinity.
Both methods leverage water’s sensitivity to changes in the biological environment. MRI excels at mapping organ- and tissue-level structures, while PIMS® reveals molecular-level interactions and signaling events—crucial for understanding how drugs work in a patient’s own biological matrix (blood, PBMCs, tissues).
Technical underpinnings

Near-Infrared Spectroscopy
- Wavelength range: 970–1940 nm
- Key principle: The absorption of NIR light by water depends on the hydrogen-bonding state of water molecules.
- High-density vs. low-density water: When proteins undergo conformational changes due to drug binding, local water molecules may transition between more “collapsed” (high-density) and more “expanded” (low-density) configurations. This shift manifests as a small but measurable change in the NIR absorption spectrum.
Label-free approach
- No fluorescent or radioactive tags: PIMS® avoids any external modifications to the drug or the sample. This ensures that native biological conditions are maintained.
- Preserves sample integrity: Without labels, there is minimal risk of altering the sample’s behavior or generating artifacts.
Instrumentation & data output
- Optical system: A specialized NIR spectrometer measures absorption changes across the relevant wavelength range.
- Dynamic spectral fingerprint: PIMS® often outputs a 3D spectral map (absorbance vs. wavelength vs. temperature/time), revealing how water resonance evolves during and after drug exposure.
- Analysis software: Automated algorithms compare baseline (untreated sample) to treatment (drug-exposed sample), quantifying the magnitude of spectral shift and identifying specific patterns indicative of drug-target engagement.
Workflow & steps
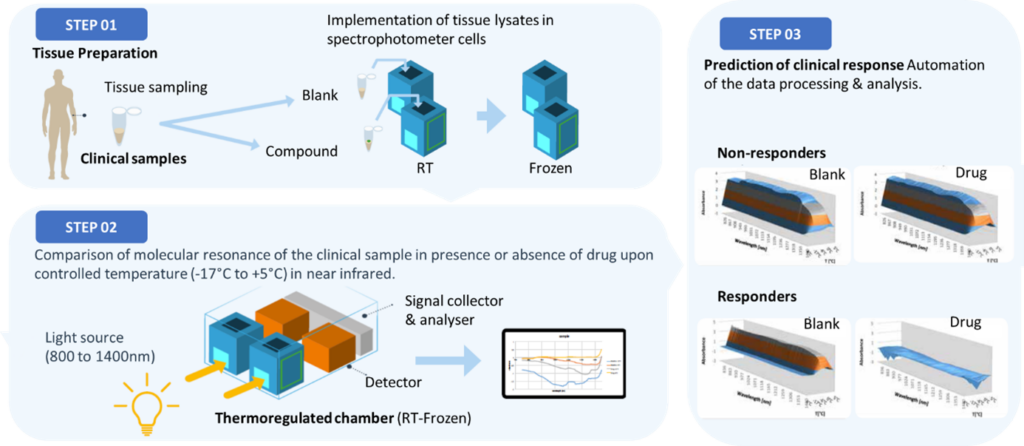
Baseline spectra
- A small volume of biological sample (e.g., blood, plasma, PBMCs, or tissue homogenate) is placed in the spectrometer.
- An initial NIR spectrum is recorded to establish the “blank” or baseline condition.
Drug challenge
- The sample is incubated with the compound of interest (e.g., a small molecule inhibitor, monoclonal antibody, or peptide).
- If the drug binds to its target protein(s), the resulting conformational changes shift the surrounding water network.
Treatment spectra
- A second NIR spectrum is recorded after the incubation period.
- By comparing baseline vs. treatment spectra, PIMS® highlights any significant shifts in water molecule resonance
Data interpretation
- 3D spectral fingerprint: The system generates a dynamic profile capturing how water absorption changes over time or temperature.
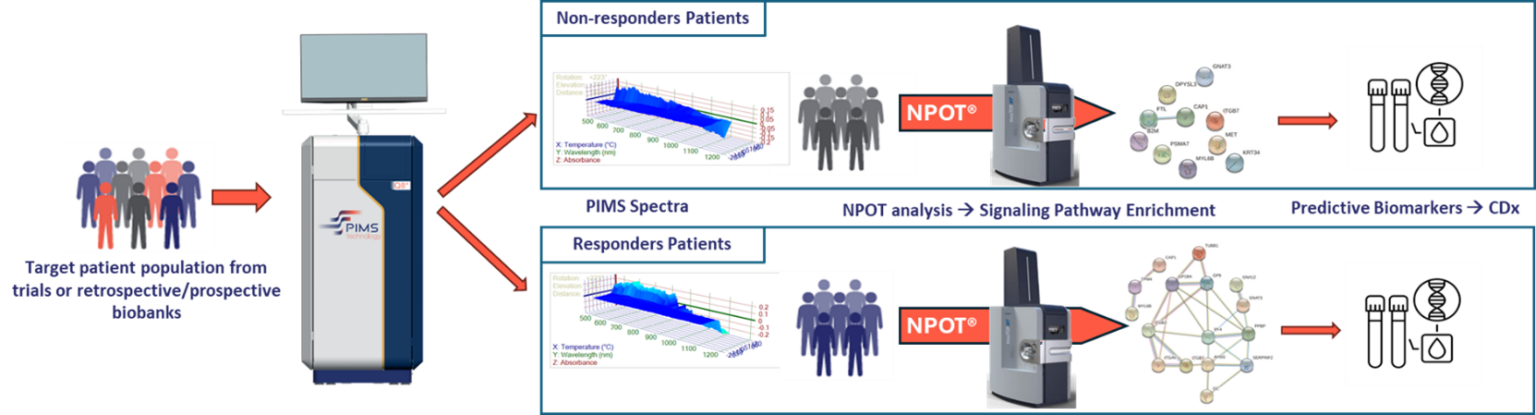
- Responder vs. non-responder: If a patient’s sample shows a strong spectral shift, it suggests meaningful drug-target interaction—predicting a likely “responder.” Minimal shift indicates a potential “non-responder.”
- Mechanistic insights: Large or distinct spectral changes may correspond to robust activation of downstream signaling pathways.
Integration with NPOT®
- Once PIMS® stratifies samples, the Nematic Protein Organization Technique (NPOT®) identifies the precise protein networks and signaling cascades involved.
- NPOT® does this by mapping out protein-protein interactions in the native, unaltered sample, pinpointing specific targets, biomarkers, or resistance mechanisms.
Applications & use cases
Drug discovery & development
- Lead optimization: Evaluate multiple candidate drugs for their interaction strengths and mechanistic effects.
- Combination therapies: Test synergy between compounds (e.g., small molecule + antibody) in a single patient sample.
Clinical trials & personalized medicine
- Patient stratification: Quickly separate likely responders from non-responders before costly or lengthy trials.
- Biomarker discovery: The combination of PIMS® and NPOT® helps identify new protein markers correlated with treatment efficacy or resistance.
Translational research
- Disease mechanisms: Gain insight into how disease-specific proteins are modulated by investigational therapies.
- Ex-vivo testing: Use patient-derived samples (e.g., tumor biopsies, PBMCs) to simulate in vivo drug response under near-physiological conditions.
Advantages of PIMS®
Holistic & non-invasive: Minimal sample preparation, preserving the integrity of the biological matrix.
High sensitivity & specificity: Detects minute shifts in water absorption, revealing subtle changes in protein conformation.
Rapid turnaround: Real-time readouts enable efficient screening and timely decision-making in drug discovery or clinical settings.
Broad compatibility: Suitable for various sample types (blood, PBMCs, tissues) and therapeutic modalities (small molecules, biologics, peptides).
Scalable to personalized medicine: Stratifies patients based on unique molecular signatures, guiding targeted therapy selection.
Future directions
Refined protocols for larger cohorts
Expanding the scope of PIMS® to bigger clinical trials for robust statistical validation.
Integration with AI/ML
Applying machine learning to spectral data could enhance predictive accuracy and uncover complex patterns.
Multi-omics approaches
Applying machine learning to spectral data could enhance predictive accuracy and uncover complex patterns.
Further clinical indications
Beyond oncology and immunology, PIMS® may benefit autoimmune disorders, metabolic diseases, and more.
Conclusion
PIMS® is transforming the way we understand and measure drug-target interactions by focusing on the most fundamental aspect of biological systems—water. Its label-free nature and near-infrared spectral readout offer unparalleled insights into molecular events that drive drug efficacy or resistance. Paired with NPOT®, PIMS® provides both predictive power and mechanistic clarity, making it a game-changer for drug discovery, clinical research, and personalized medicine.