Abstract
Background
Inflammatory Bowel Disease (IBD), including Crohn’s disease and ulcerative colitis, is a chronic condition that significantly impacts patients’ quality of life. While biologic therapies like anti-TNF agents have revolutionized treatment, a substantial proportion of patients do not respond to these therapies. Vedolizumab, an anti-integrin antibody, offers an alternative for patients refractory to anti-TNF treatments. However, predicting which patients will respond to vedolizumab remains a challenge, highlighting the need for personalized medicine approaches.
In this case study, we demonstrate how Physiological Intermolecular Modulation Spectroscopy (PIMS®), a patented label-free technology, was used to predict response to vedolizumab in anti-TNF refractory IBD patients with high accuracy. This study, conducted in collaboration with the University of Erlangen-Nürnberg, Germany, and with Takeda Pharmaceuticals Gmbh, showcases the potential of PIMS® in guiding personalized treatment decisions.
Objective
The study aimed to:
- Predict clinical response to vedolizumab in anti-TNF refractory IBD patients using PIMS®.
- Identify underlying molecular networks associated with response using the Nematic Protein Organization Technic (NPOT®).
Methods
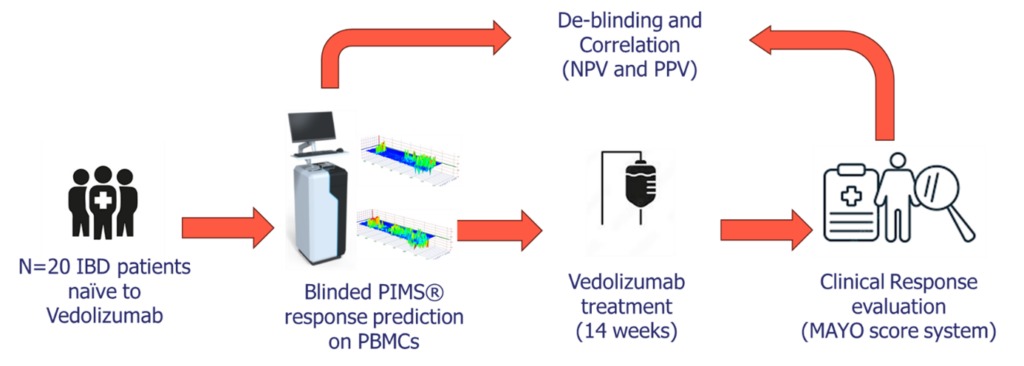
Patient cohort:
- 20 IBD patients (13 Crohn’s disease, 7 ulcerative colitis) who had previously failed at least one anti-TNF therapy were enrolled.
- Blood samples were collected at baseline (week 0) and week 14 of vedolizumab therapy.
PIMS® analysis:
- Peripheral blood mononuclear cells (PBMCs) were isolated and analyzed using PIMS®.
- PIMS® measured changes in water molecule resonance and macromolecular conformation in response to vedolizumab, generating dynamic fingerprints of molecular interactions.
- The Individual Macromolecular Volume (IMV) was calculated to differentiate responders from non-responders.
NPOT® analysis:
- NPOT® was used to identify functional molecular networks in responders by analyzing protein-protein interactions in PBMCs.
- Proteins were identified using liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Clinical response assessment:
- Response was defined as a reduction in disease activity scores (Harvey-Bradshaw Index for Crohn’s disease and partial Mayo Score for ulcerative colitis) at week 14.
Results
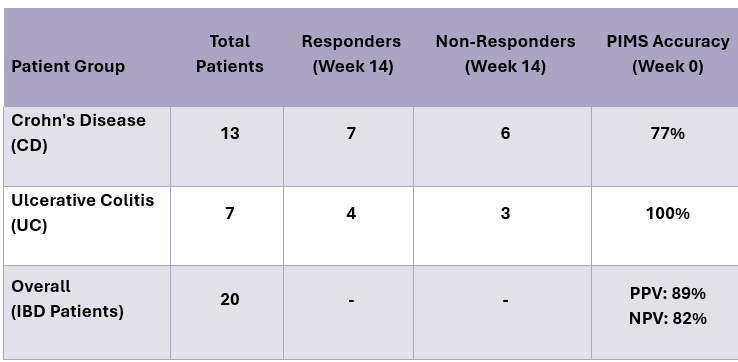
PIMS® predicts response with high accuracy:
- PIMS® accurately predicted clinical response to vedolizumab in 100% of ulcerative colitis patients and 77% of Crohn’s disease patients.
- Overall, PIMS® achieved an 89% positive predictive value (PPV) and an 82% negative predictive value (NPV) for all IBD patients.
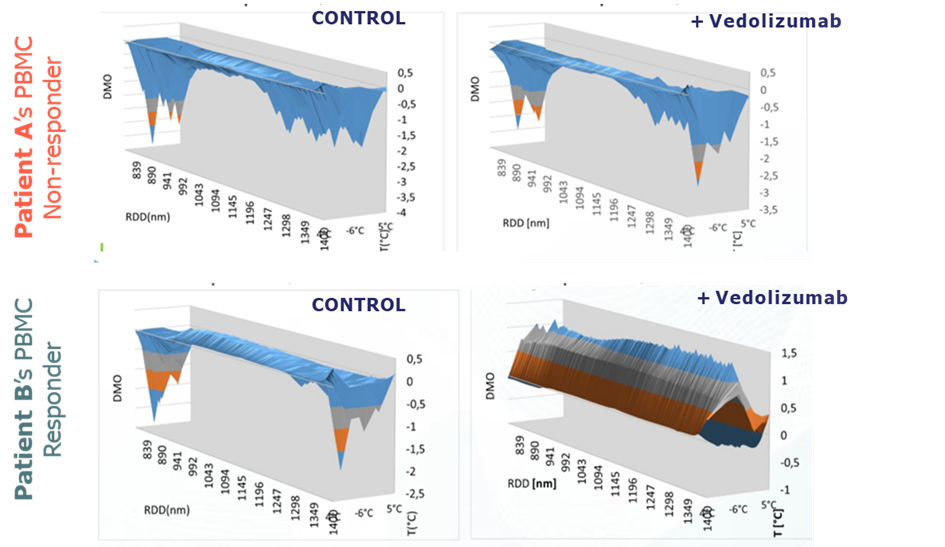
3D spectra profiles generated by PIMS® instrument. Individual Macromolecular Volume (IMV) profiles from Patient A and Patient B are generated from the continuous sampling of NIR spectra of patients PBMC homogenates as the temperature rises from −17 to 5°C. Control panels: PIMS® analysis without compound (baseline). + Vedolizumab panels: the same patient’s PBMCs incubated with vedolizumab. In non-responder patient A, there is no change in IMV in presence of vedolizumab. Responder patient: Vedolizumab increased considerably the IMV. DMO, differential molecular oscillation; RDD, relative dynamic diffraction.
NPOT® reveals functional molecular networks:
- In both responders and non-responders, NPOT® identified the primary target of Vedolizumab, α4β7 integrin (ITGB7).
- In responders, NPOT® identified a distinct molecular network involving co-receptors of ITGB7 such as , ITGAV, ITGB3 proteins such as , PF4, and AHSG, which are associated with vedolizumab’s mechanism of action. In Particular,
- Non-responders lacked this functional network, explaining their lack of response to therapy.
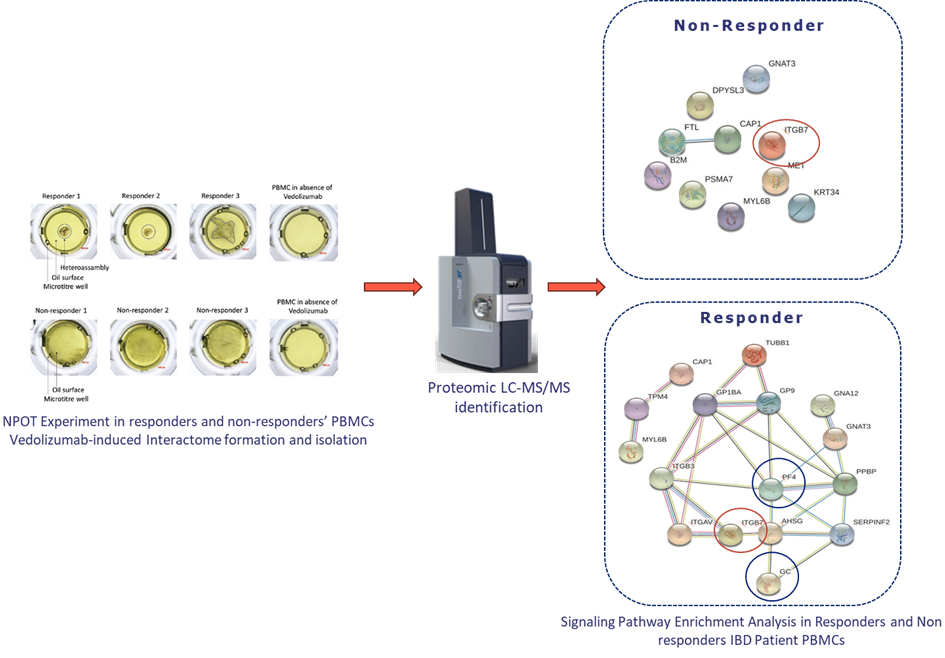
Key biomarkers identified:
Platelet Factor 4 (PF4) and Vitamin D-binding protein (GC) emerged as potential biomarkers for predicting vedolizumab response, offering insights into the molecular basis of treatment efficacy.
Discussion and conclusion
Discussion
This study highlights the power of PIMS® in personalized medicine for IBD. By analyzing molecular interactions in patient blood samples, PIMS® can stratify patients into responders and non-responders before initiating therapy, enabling more informed treatment decisions. The integration of NPOT® further elucidated the underlying molecular networks, providing mechanistic insights into vedolizumab’s action.
The identification of PF4 and GC as potential biomarkers opens new avenues for developing companion diagnostics, which could further refine patient selection for vedolizumab therapy.
Conclusion
PIMS® technology offers a non-invasive, rapid, and highly accurate method for predicting response to vedolizumab in anti-TNF refractory IBD patients. By combining PIMS® with NPOT®, clinicians can not only predict treatment outcomes but also gain insights into the molecular mechanisms driving response, paving the way for truly personalized medicine in IBD.
Why this matters
For patients
PIMS® ensures that patients receive the most effective treatment from the start, reducing unnecessary side effects and improving quality of life.
For clinicians
PIMS® provides a reliable tool for treatment decision-making, enhancing the precision of IBD management.
For researchers
The integration of PIMS® and NPOT® offers a powerful platform for biomarker discovery and mechanistic studies in drug development.